HYDROXYUREA Synthesis, SAR, MCQ,Structure,Chemical Properties and Therapeutic Uses
Hydroxyurea
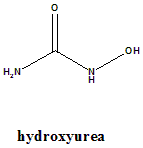
IUPAC nomenclature
Hydroxyurea
Classification
Hydroxurea falls under the category of Antineoplastic cytotoxic drug. [1]
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 76.055 g/mol |
| 2 | Appearance | White to off-white crystalline solid |
| 3 | Melting point | 141°C |
| 4 | Solubility | 13.15M in water |
| 5 | Octanol/Water partition coefficient | -1.8 |
| 5 | Presence of ring | No ring structure present |
Mechanism of Action
Hydroxyurea transported into the cells after getting converted to nitroxide in vivo. At the active site of M2 protien subunit of ribnucleotide reductase, it quenches the tyrosyl free radical, thus, the enzyme got deactivated. DNA synthesis is selectively inhibited which leads to the death of cell at in the S phase. The repair of the DNA is also inhibited by the hydroxyurea. Increase in the level of fetal hemoglobin due to activation of soluble guanylyl cyclase by hydroxyurea-derived nitrooxide. [2]
Structural Activity Relationship
- Hydroxyl group is necessary for the inhibition of DNA synthesis.
- 1-methyl and 1-ethyl hydroxyurea are highly antimitotic.
- Carbonyl group substitution at 1 position decreases the antimitotic activity of drug.
- Substitution of the hydroxyl proton with a methyl group converts the drug into an inactive form.
- Antimitotic property persistent if the hydroxyl proton is substituted by carbonyl group.
- All the di- and tri- substituted derivatives with methyl and n-butyl group in rthe hydroxyl proton are inactive.
- Tetra hydroxyl urea is highly lipid soluble. [3]
Methods of Synthesis
Hydroxyurea can be synthesized by the interaction of hydroxylamine hydrochloride and potassium cyanide.

Therapeutic Uses
Hydroxyurea is used for the treatment of:
- Refractory ovarian cancer
- Melanoma
- Head and neck cancer
- Essential thrombocytosis
- Polycythemia vera
- Chronic myeloid leukemia
Side Effects
- Low blood count is the common side effect of the drug.
- Some people may suffer from side effects like loss of hair,
- Darkening of the skin, discoloration of the skin, thickening of nails, loss of appetite, mouth sores, diarrhea, nausea and vomiting.
MCQs
Q.1 Hydroxyurea is the IUPAC nomenclature of which drug?
a) Anastrozole
b) Exemestane
c) Imitinab
d) Hydroxyurea
Q.2 Predict the incorrect statement related to the therapeutic uses of drug Hydroxyurea-
a) It is used for the treatment of melanomas
b) It is used for the treatment of refractory ovarian cancer
c) It is used for the treatment of mouth sores
d) It is used for the treatment of head and neck cancers
Q.3 Match the following with respect to the SAR of drug Hydroxyurea
| i. Hydroxyl group | A. Necessary for the activity of drug |
| ii. Replacement of proton of hydroxyl group with methyl group will | B. Is not necessary for the activity of drug |
| C. Increase the activity of drug | |
| D. Decrease the activity of drug |
a) i-A, ii-C
b) i-A, ii-D
c) i-B, ii-C
d) i-B, ii-D
Q.4 Which amongst the following drugs shows its effect through inhibition of ribonuceloside reductase enzyme?
a) Hydroxyurea
b) Actinomycin D
c) Nafarelin
d) Bicalutamide
Q.5 Hydroxyurea drug belongs to which class?
a) Antibodies
b) Glucocorticoids
c) Immunosuppressant drug
d) Antineoplastic cytotoxic drug
Q.6 Which of the following is not a side effect of Hydroxyurea?
a) Loss of hair
b) Discoloration of skin and nails
c) Darkening of the skin
d) None of the above
Q.7 Which amongst the following drugs is having least number of ring system in its structure-
a) Morphine
b) Cyclophosphamide
c) Hydroxyurea
d) All have same number of ring structures
ANSWERS
1-d
2-c
3-b
4-a
5-d
6-d
7-c
REFERENCES
[1] Tripathi KD. Essentials of Medical Pharmacology, 6thEdn. Jaypee Brothers Medical Publishers (P) Ltd. 2008: 820. [2] Yarbro JW. Mechanism of action of hydroxyurea. InSeminars in oncology 1992 Jun (Vol. 19, No. 3 Suppl 9, pp. 1-10). [3] Ruey JY, Van Scott EJ. Antimitotic Effects of Hydroxyurea and Its Derivatives: Structure-Activity Relationships. Journal of Investigative Dermatology. 1974 Sep 1;63(3):279-83.