METIPRANOLOL Synthesis, SAR, MCQ,Structure,Chemical Properties and Therapeutic Uses
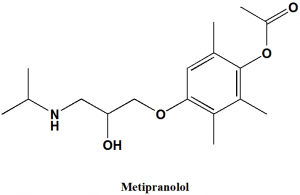
Metipranolol
IUPAC nomenclature
(RS)-4-{[-2-hydroxy-3-(isopropylamino)propyl]oxy}-2,3,6-trimethylphenyl acetate.
Classification
Metipranolol is a nonselective ß-adrenergic antagonist.
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 309.4 g/mol |
| 2 | Physical appearance | Present in solid form. |
| 3 | Melting point | 106°C |
| 4 | Solubility | 589 mg/L |
| 5 | Octanol/water partition coefficient | 2.66 |
| 6 | Presence of ring | Benzene |
| 7 | Number of chiral centers | 1 |
Mechanism of Action
- Metipranolol bind with ß-adrenergic and blocks it.
- It has weak local anesthetic and myocardial depressant activity.
- When used in ophthalmic, it reduces aqueous humor production. Aqueous humor outflow may also take place.
Structure Activity Relationship
- Increasing the chain length of the side chain prevents appropriate binding of the required functional groups to the same receptors side.
- Side chain of aryloxypropanolamines can adopt a conformation that places the hydroxyl and amine groups into approximately the same position in space.
- Aryloxypropalonamines permits a close overlap with the arylethanomine side chain.
- Aryloxypropanolamines are more potent than aryloxyethanolamines. [1]
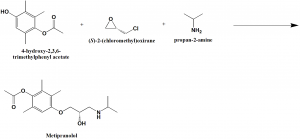
Method of synthesis
4-hydroxy-2,3,6-trimethylphenyl acetate; (S)-2-(chloromethyl)oxirane and propan-2-amine reacts together to produce metipranolol.
Therapeutic Uses
Metipranolol is used for treatment of:
- Glaucoma
- Ocular hypertension
Side Effects
Side effects of metipranolol are:
- Swelling and discomfort around the eyes
- Bronchospasm
- Sudden numbness and weakness
- Headache
- Confusion
- Slow heart rate
- Shortness of breath
- Weight gain
- Watering of eyes
- Blurred vision
- Insomia
- Altered sense of taste or smell
MCQs
Q.1 Match the following with correct SAR of the drug metipranolol-
| i. Increasing side chain length | A. Prevents appropriate binding of the required functional groups to the same receptors side. |
| ii. Aryloxypropanolamines are | B. Stimulates appropriate binding of the required functional groups to the same receptors side |
| C. Less potent than aryloxyethanolamines | |
| D. More potent than aryoxyethanolamines |
a) i-A, ii-C
b) i-A, ii-D
c) i-B, ii-C
d) i-B, ii-D
Q.2 Correct sequence for the True/False for correct IUPAC names of the drug can be?
- Metipranolol: (RS)-4-{[-2-hydroxy-3-(isopropylamino)propyl]oxy}-2,3,6-trimethylphenyl acetate.
- Esmolol: 3-(isopropylamino)propan-2-ol
- Bisprolol: (RS)-1-{4-[(2-Isopropoxyethoxy)methyl]phenoxybenzamide
- Metoprolol: (RS)-1-[4-(2-Methoxyethyl)phenoxy]-3-[(propan-2-yl)amino]propan-2-ol
a) TFFT
b) TFTF
c) TTFF
d) TTFT
Q.3 Number of chiral centers present in the structure of metipranolol?
a) 0
b) 1
c) 2
d) 3
Q.4 Mechanism of action of metipranolol is based on?
a) Blocking of α1-adrenergic receptors
b) α1-adrenergic agonism
c) ß-adrenergic antagonism
d) None of these
Q.5 Which amongst the following is not a therapeutic use of drug metipranolol?
a) Glaucoma
b) Ocular hypertension
c) Osteoporosis
d) None of these
Q.6 Which of the following drug and their classification are correct?
I. Metipranolol: nonselecvtive ß-adrenergic antagonist
II. Metoprolol: selective ß1-adrenergic antagonist
III. Amphetamine: nonselective α-adrenergic agonist
IV. Clonidine: Mixed acting sympathomimetics
a) I, III
b) I, II
c) II, III, IV
d) I, II, III, IV
Q.7 Octanol/water partition coefficient of metipranolol is?
a) 2.66
b) 1.2
c) -5.4
d) 5.7
ANSWERS
1-b
2-a
3-b
4-c
5-c
6-b
7-a