POLYTHIAZIDE Synthesis, SAR, MCQ,Structure,Chemical Properties and Therapeutic Uses
Polythiazide
IUPAC nomenclature
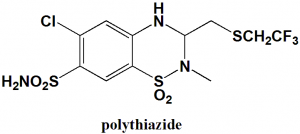
6-chloro-2-methyl-3-{[(2,2,2-trifluoroethyl)thio]methyl}-3,4-dihydro-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide
Classification
- Thiazide diuretic
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 439.9 g/mol |
| 2 | Physical appearance | White crystalline powder |
| 3 | Melting point | 214oC |
| 4 | Solubility | Soluble in methanol, acetone and dimethylformamide; practically insoluble in water |
| 5 | Octanol/water partition coefficient | 1.9 |
| 5 | Presence of ring | Benzothiazine |
| 6 | Number of chiral centers | Not present |
Mechanism of Action
- Polythiazide prevents active chloride reabsorption at the early distal tubule through the sodium chloride contransportor which results in an increase in the excretion of sodium, chloride and water from the body.
- The drug also binds with thiazide-sensitive Na-Cl transportor and prevents sodium ion transport across the renal tubular epithelium. This increases the potassium excretion through Na-K exchange mechanism.
- It can also mediate its actions on carbonic anhydrase in the smooth muscle or on the large-conductance KCa channel found in smooth muscle.
Structure Activity Relationship
General structure activity of thiazide diuretics can be summarized as:
- Chlorothiazide is the simplest member of the series.
- Hydrogen atom at the 2-N is most acidic due to presence of electron-withdrawing group.
- Sulfonamide group at C-7 position provides additional acidity to the drug.
- Electron withdrawing group is essential at position 6 for diuretic activity of the drug.
- Substitution oh hydrogen at 6 position gives little diuretic activity, whereas, substitution with chloro and trifluoromethyl groups gives highly active compounds.
- Substitution of electron donating group at position 6 significantly reduces the diuretic activity.
- Replacement or removal of sulfonamide groups from position 7 significantly reduces the diuretic activity.
- Saturation of the double bond to give 2,4-dihydro derivative are 10-folds more active than the unsaturated compounds.
- Substitution of a lipophillic group at 3 position increases the potency.
- Substitution with the entities such as haloalkyl, aralkyl or thioether gives compounds with longer duration of action due to increased lipid solubility.
- Alkyl substitution at the 2-N position can increase the action duration. [1]
Method of synthesis
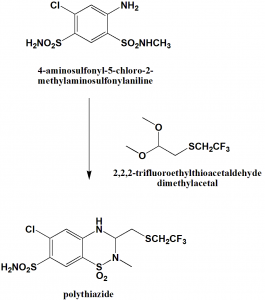
.Condensation of 4-aminosulfonyl-5-chloro-2-methyl-aminosulfonylaniline with the help of 2,2,2-trifluoroethylthioacetaldehyde dimethylacetal produces polythiazide drug.
Medicinal Uses
Polythiazide is used for:
- Reducing edema
- Decreasing blood pressure
Side Effects
Side effects of polythiazide are:
- Nausea
- Blurred vision
- Vomiting
- Dizziness
- Fatigue
- Diarrhea
- Low blood sodium and potassium levels
- Loss of appetite
- Stomach cramps
- Photosensitivity
- Muscle cramps
- Fainting
- Seizures
MCQs
Q.1 “6-chloro-2-methyl-3-{[(2,2,2-trifluoroethyl)thio]methyl}-3,4-dihydro-2H-1,2,4-benzothiadiazine-7-sulfonamide 1,1-dioxide” is the IUPAC nomenclature of which drug?
a) Polythiazide
b) Atropine
c) Amlodipine
d) Tropicamide
Q.2 Melting point of drug Polythiazide is?
a) 408.9oC
b) 214 oC
c) 345 oC
d) 741 oC
Q.3 Match the following with correct classifications of the drugs.
| i. Polythiazide | A. Narcotic antagonist |
| ii. Phensuximide | B. Anticonvulsant |
| iii. Pentobarbital | C. Sedative-hypnotics |
| iv. Nalorphine | D. Calcium channel blocker |
a) i-C, ii-A, iii-D, iv-B
b) i-D, ii-C, iii-A, iv-B
c) i-D, ii-B, iii-C, iv-A
d) i-C, ii-D, iii-B, iv-A
Q.4 Mechanism of action of the drug Polythiazide includes?
I. Prevention of active chloride reabsorption.
II. Binding with thiazide-sensitive Na-Cl transportor
III. Increase in excretion of water from the body.
a) I, II, III
b) II, III
c) I, III
d) I, II
Q.5 Correct sequence for True and False for the given statements related with the SAR of Thiazide diuretics can be?
- Electron withdrawing group is essential at position 6 for diuretic activity of the drug.
- Substitution oh hydrogen at 6 position gives little diuretic activity, whereas, substitution with chloro and trifluoromethyl groups gives highly active compounds.
- Substitution of electron donating group at position 6 significantly increases the diuretic activity
a) TFF
b) FFT
c) TTF
d) FFF
Q.6 Number of chiral carbons present in the structure of polythiazide is?
a) 0
b) 1
c) 2
d) 3
Q.7 The drug Polythiazide is mainly used for?
a) Reducing edema
b) Treatment of hypotension
c) Treatment of asthma
d) All of the above
Participate in Online FREE GPAT TEST: CLICK HERE
Participate in Online FREE Pharmacist TEST: CLICK HERE
Participate in Online FREE Drug Inspector TEST: CLICK HERE
Participate in CSIR NET JRF Mock Test
ANSWERS
1-a
2-b
3-c
4-a
5-c
6-a
7-a