ESTAZOLAM Synthesis, SAR, MCQ,Structure,Chemical Properties and Therapeutic Uses
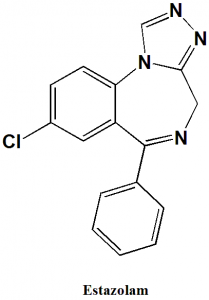
Estazolam
IUPAC nomenclature
8-chloro-6-phenyl-4H-[1,2,4]triazolo[4,3-a][1,4]benzodiazepine.
Classification
Estazolam is a benzodiazepine sedative-hypnotic.
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 294.74 g/mol |
| 2 | Physical appearance | Solid |
| 3 | Melting point | 228.5°C |
| 4 | Octanol/water partition coefficient | 4.7 |
| 5 | Solubility | Practically insoluble |
| 6 | Presence of ring | Diazepine, triazole, benzene |
| 7 | Number of chiral centers | Not present |
Mechanism of Action
i. Estazolam binds nonspecifically with benzodiazepine receptors BNZ1.
ii. It coupled with GABAA receptors and increases the GABA affinity for the GABA receptor.
iii. This results in opening of the chloride channel and thus, causes hyperpolarization of the cell membrane which prevents further excitation of the cell.
Structure Activity Relationship
- Ring A should include an aromatic or heteroaromatic ring for binding with 5-phenyl-1,4-benzodiazepin-2-one derivatives.
- An electronegative group at 7-position of the ring A increases the functional anxiolytic activity.
- Substitutions at 6, 8 or 9 position with electronegative group on ring A will decrease the functional anxiolytic activity.
- When Heterocycles used as ring A, drug shows poor pharmacological activity.
- A proton-accepting group is essential on Ring B for binding with GABAA
- When the proton accepting group is present on the 2-position of the ring B, and is in coplanar spatial orientation with Ring A, maximum activity is observed.
- Replacement of oxygen with sulfur in ring B results in alteration in the selectivity for binding with GABA BZR subpopulations, but anxiolytic properties are maintained.
- There is no effect on agonist activity, but the antagonist activity decreases when methylene 3-position or imine nitrogen of the ring B is substituted.
- Derivatives having the 3-hydroxy moiety are fast excreted.
- Sterically large substituents on ring B, like tert-butyl group reduces the receptor affinity and the in vivo activity.
- 4,5-double bond and 4-position nitrogen is not essential for anxiolystic activity.
- BZR affinity is decreased if C=N bond is replaced with C-N bond.
- 5-phenyl ring C is not necessary for the binding with BZR.
- Substitution at the para position of the ring C decreases the agonist activity of the drug.
- There is no change observed in the agonist property of the drug when there is substitution at ortho position.
- When 1,2-bond f the ring C is annelated with an additional electron rich ring such as imidazole, affinity of the BZR increases. [1]
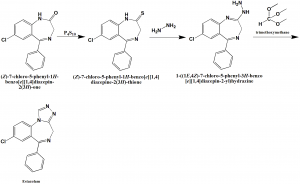
Method of synthesis
i. (Z)-7-chloro-5-phenyl-1H-benzo[e][1,4]diazepin-2(3H)-one reacts with phosphorus pentasulfide to give (Z)-7-chloro-5-phenyl-1H-benzo[e][1,4] diazepine-2(3H)-thione .
ii. The compound then undergoes reaction with hydrazine to form 1-((1E,4Z)-7-chloro-5-phenyl-3H-benzo [e][1,4]diazepin-2-yl)hydrazine.
iii. On reaction with trimethoxyerhane, Estazolam is synthesized.
Therapeutic Uses
Estazolam is used for:
- Treatment of insomnia
Side Effects
Side effects of Estazolam are:
- Dizziness
- Memory loss
- Hallucinations
- Confusion
- Mood changes
- Anxiety
- Aggressive behavior
- Loss of coordination
MCQs
Q.1 Sedative-hypnotic effect of estazolam is through?
a) Increase the affinity of GABA for GABA receptors
b) Decrease the affinity of GABA for GABA receptors
c) Blocking the calcium channel
d) Alkalization of nuclear material
Q.2 Therapeutic use of drug Estazolam is/are?
a) Treatment of memory loss
b) Sedation during time of Insomnia
c) Treatment of Diabetes
d) Both a) and b)
Q.3 Which amongst the following are the correct statements with respect to the SAR of drug estazolam?
I. Tri-keto form is least stable in aqueous solution.
II. 4,6-dialcoholic tautomeric forms are most stable in aqueous solution.
III. 5,5-disubstituted barbituric acid is the prime requirement for the barbituares to be sedative hypnotics.
a) I, II, III
b) I, II
c) II, III
d) III
Q.4 Number of chiral centers present in the structure of Estazolam?
a) 0
b) 1
c) 2
d) 3
Q.5 Correct sequence for the True/False for the physiochemical properties of the drug Estazolam?
I. Molecular weight = 294.74 gm/mol
II. It is insoluble in water
III. Melting point is 174°C
a) TTF
b) FTF
c) TFT
d) FTT
Q.6 Correct statements for the IUPAC nomenclatures of the drugs are?
I. Estazolam: 5-Ethyl-5-phenyl-1,3-diazinane-2,4,6-trione
II. Oxazepam: 5-oxo-D-prolyl-L-histidyl-Ltryptophyl-L-seryl-Ltyrosyl-3-(1H-indol-2-yl)-L-alanylleucyl-L-arginyl-L-prolylglycinamide.
III. Epinephrine: (R)-3-[-1-hydroxy-2-(methylamino)ethyl]phenol
IV. Zaleplon: (RS)-N-[4-cyano-3-(trifluoromethyl)phenyl]-3-[(4-fluorophenyl)sulfonyl]-2-hydroxy-2-methylpropanamide.
a) I, II, III
b) I, IV
c) I, III
d) None
Q.7 Match the following drugs with their correct classifications-
| i. Oxazepam | A. Benzodiazepine |
| ii. Clozapine | B. Nonbenzodiazepine agonist |
| iii. Zolpidem | C. Benzapene |
| iv. Butabarbital | D. Barbiturate |
a) i-D, ii-B, iii-C, iv-A
b) i-D, ii-C, iii-B, iv-A
c) i-A, ii-C, iii-B, iv-D
d) i-A, ii-C, iii-B, iv-D
Participate in Online FREE GPAT TEST: CLICK HERE
Participate in Online FREE Pharmacist TEST: CLICK HERE
Participate in Online FREE Drug Inspector TEST: CLICK HERE
ANSWERS
1-a
2-b
3-d
4-a
5-a
6-d
7-c
REFERENCES
[1] Lemke TL, Zito SW, Roche VF, Williams DA. Essentials of Foye’s principles of medicinal chemistry. Wolters Kluwer; 2017, 473-474.