PENTOBARBITAL Synthesis, SAR, MCQ,Structure,Chemical Properties and Therapeutic Uses
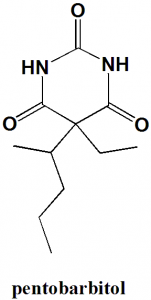
Pentobarbital
IUPAC nomenclature
5-Ethyl-5-(1-methylbutyl)-2,4,6(1H,3H,5H)-pyrimidinetrione
Classification
Pentobarbital is a barbiturate sedative-hypnotic.
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 226.27 g/mol |
| 2 | Physical appearance | Crystalline granules or white powder |
| 3 | Melting point | 129.5°C |
| 4 | Octanol/water partition coefficient | 2.1 |
| 5 | Solubility | Slightly soluble in water; soluble in ethyl alcohol |
| 6 | Presence of ring | Pyrimidine |
| 7 | Number of chiral centers | 1 |
Mechanism of Action
- Pentobarbital increases the duration of time for which the chloride ionopore is open through binding with GABAA
- The post-synaptic inhibitory effect of thalamus prolongs.
- There is decreases in GABA sensitive neuronal calcium conductance.
- The overall result is acute potentiation of inhibitory GABAnergic tone.
- Drug also inhibits excitoryAMPA-type glutamate receptors which results in profound suppression of glutamatergic neurotransmission.
Structure Activity Relationship
- Tri-keto form is most stable in aqueous solution.
- 4,6-dialcoholic tautomeric forms are least stable in aqueous solution.
- 5,5-disubstituted barbituric acid is the prime requirement for the barbituares to be sedative hypnotics.
- Esterification of either of the 1,3-diazine nitrogens decreases hypnotic activity.
- Substitution of either of the 1,3-diazine nitrogens with aliphatic carbons retains the anticonvulsive properties.
- Esterification of the 5th-position substituents yields agents with analgesic activity but with weak hypnotic properties.
- Introduction of the polar functional group at the 5th– position yields compounds which are fully devoid of sedative-hypnotic or anticonvulsive activity.
- As the number of carbons at R2 carbon increases, the lipophillicity of the drug increases.
- Modification of the 2nd-position oxygen of the barbiturate backbone with sulfur atom yields thiobarbiturate derivatives with increased lipophillicity, shorter duration of action, faster time of onset compared to oxy-derivative. [1]
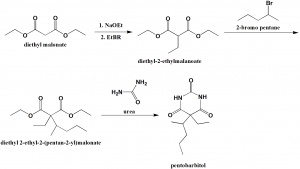
Method of synthesis
i. Diethyl malonate reacts with sodium ethoxide followed by reaction with ethylbromide to give diethyl 2-ethylmalonate.
ii. The compound undergoes reaction with 2-bromopentane.
iii. The above formed compound on reaction with urea yields pentobarbital. [2]
Therapeutic Uses
Pentobarbital is used for:
- Treatment of insomnia
- Sedative before surgery
- Emergency Treatment of seizures
Side Effects
Side effects of Pentobarbital are:
- Insomnia
- Dizziness
- Drowsiness
- Loss of coordination
- Nausea
- Vomiting
- Constipation
- Nightmares
- Restlessness
MCQs
Q.1 Correct statements related with the physicochemical properties of drug Pentobarbital?
I. Molecular weight = 226.27 gm/mol
II. Physical appearance: red-brown crystalline powder
III. Melting point: 402.5K
IV. Triazole ring present
a) I, III
b) II, IV
c) I, II
d) I, II, IV
Q.2 Match the following of the drugs with their correct IUPAC names.
| i. Pentobarbital | A. 5-Ethyl-5-(1-methylbutyl)-2,4,6(1H,3H,5H)-pyrimidinetrione |
| ii. Midazolam | B. N,N-Dimethyl-2-[6-methyl-2-(4-methylphenyl)imidazo[1,2-a]pyridin-3-yl]acetamide hemitartrate
|
| iii. Zolpidem | C. 8-chloro-6-(2-fluorophenyl)-1-methyl-4H-imidazo[1,5-a][1,4]benzodiazepine |
| iv. Alprazolam | D. 8-Chloro-1-methyl-6-phenyl-4H-[1,2,4]triazolo[4,3-a] [1,4]benzodiazepine |
a) i-B, ii-C, iii-A, iv-D
b) i-A, ii-C, iii-B, iv-D
c) i-D, ii-A, iii-C, iv-B
d) i-B, ii-D, iii-C, iv-A
Q.3 Correct steps for the mechanism of action of the drug Pentobarbital?
I. Binds with GABAA receptors
II. Increase in GABA affinity for GABA receptors
III. Opening of chloride channel
IV. Hyperpolarisation
a) I – II – III – IV
b) II – I – III – IV
c) III – I – II – IV
d) II – IV – III – I
Q.4 Correct sequence for True/false for the classification of the drug can be?
- Pentobarbital: benzodiazepine sedative-hypnotic
- Quetipine : benzodiazepine sedative-hypnotic
- Flurazepam: benzodiazepine
- Eszopiclone: butyrophenone
a) TFTF
b) FFTF
c) TFFF
d) FTFT
Q.5 Triketo form of pentobarbital is?
a) Most stable in aqueous solution
b) Least stable in aqueous solution
c) Most soluble in organic solvent
d) Least soluble in organic solvent
Q.6 The correct sequence for the steps for synthesis of drug pentobarbital from diethyl malanoate is?
I. Reaction with 2-bromopentane
II. Reaction with sodium ethoxide
III. Condensation with urea.
a) III – I – II
b) I – II – III
c) I – III – II
d) II – I – III
Q.7 Side effect of drug Pentbarbital?
a) Insomnia
b) Loss of coordination
c) Nightmares
d) All of the above
Participate in Online FREE GPAT TEST: CLICK HERE
Participate in Online FREE Pharmacist TEST: CLICK HERE
Participate in Online FREE Drug Inspector TEST: CLICK HERE
ANSWERS
1-a
2-b
3-a
4-c
5-a
6-d
7-d
REFERENCES
[1] Lemke TL, Zito SW, Roche VF, Williams DA. Essentials of Foye’s principles of medicinal chemistry. Wolters Kluwer; 2017, 490-91 [2] Vardanyan R, Hruby V. Synthesis of essential drugs. Elsevier; 2006 Mar 10..