FENTANYL Synthesis, SAR, MCQ,Structure,Chemical Properties and Therapeutic Uses
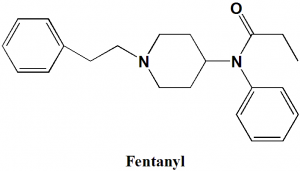
Fentanyl
IUPAC nomenclature
N-phenyl-N-[1-(2-phenylethyl)piperidin-4-yl]propanamide
Classification
- Fentanyl falls under category of opioid analgesics.
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 336.5 g/mol |
| 2 | Physical appearance | Solid crystalline powder |
| 3 | Melting point | 87.5°C |
| 4 | Solubility | 200 mg/L in water |
| 5 | Octanol/water partition coefficient | 4.05 |
| 6 | Presence of ring | Piperidine, phenyl |
| 7 | Number of chiral centers | Not present |
Mechanism of Action
- Fentanyl binds with the mu opioid receptors which are coupled with G-proteins.
- This result in activation of the opioid receptors causes GTP to be exchanged for GDP on the G-protein.
- This further down regulates adenylate cyclase, which in turn reduces the cAMP concentration.
- cAMP dependent influx of calcium ions into the cell is reduced.
- Inhibition of the nerve activity is due to hyperpolarization of the cell because of exchange of GTP for GDP.
Structure Activity Relationship
SAR for Opiates can be summarized as follows:
- Replacement of phenolic hydroxyl into –OCH3/-OC2H5 will make the compound less analgesic and cough suppression will also takes place.
- Replacement of alcoholic hydroxyl with –OCH3 makes the compound 5 times more active.
- Replacement of alcoholic hydroxyl with -OC2H5 makes the compound 2.4 times more active than morphine.
- Replacement of alcoholic hydroxyl with –OCOCH3 will also activates the compound by 4.2 times.
- Replacement of alcoholic hydroxyl with ketone group inactivates the compound and makes it lesser active.
- By hydrogenation of alicyclic unsaturated linkage, activity increases by 1.2 times.
- On replacement of the methyl group from tertiary nitrogen by hydrogen atom, activity decreases.
- On replacement of N-CH3 by NCH2CH2Ph, activity increases by 14 times.
- When the methyl group of tertiary nitrogen replaced by N-allyl/methallyl/propyl, the compound so formed acts like the Morphine antagonist.
- When the methyl group of tertiary nitrogen replaced by N(CH3)2 Cl– , compound have curare action and it do not possesses any analgesic activity.
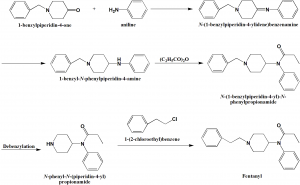
Method of synthesis
i. 1-benzylpiperidin-4-one is condensed with aniline to give corresponding Schiff’s base.
ii. Double bond of the Schiff base is reduced by lithium aluminum hydride to give 1-benzyl-4-anilinopiperidine.
iii. Product is then acylated with propionic acid anhydride to poduce –benzyl-4-N-propinoylanilinopiperidine.
iv. Above formed compound undergoes debenzylation using hydrogen and palladium on carbon catalyst to give 4-N-propanoylanilinopiperidine.
v. Last formed compound is N-alkylated with 2-phenylethylchloride to produce fentanyl. [1]
Therapeutic Uses
Fentanyl is used for:
- Anesthesia
- Treatment of pain
Side Effects
Side effects of fentanyl are:
- Diarrhea
- Nausea
- Dry mouth
- Constipation
- Somnolence
- Confusion
- Dry mouth
- Sweating
- Asthenia
- Abdominal pain
- Headache
- Fatigue
- Anorexia
- Weight loss
- Dizziness
- Nervousness
- Apnoea
- Hypoventilation
- Shortness of breath
- Anxiety
- Depression
- Hallucinations
- Urinary retention
MCQs
Q.1 What can be the correct IUPAC nomenclature of Fentanyl?
a) (5α,6α)-7-hydro-4,5-epoxy-3-ethoxy-17-methylmorphinan-6-ol
b) N-phenyl-N-[1-(2-phenylethyl)piperidin-4-yl]propanamide
c) N-[1-(2-phenylethyl)piperidin-4-yl]propanamide
d) 2-chloro-1,1-difluoro-1-methoxyethane
Q.2 Which amongst the following statements is/are incorrect related to the SAR of opiates?
I. Replacement of alcoholic hydroxyl with -OC2H5 makes the compound 2.4 times more active than morphine.
II. Replacement of alcoholic hydroxyl with –OCOCH3 will also activates the compound by 4.2 times.
III. Replacement of alcoholic hydroxyl with ketone group inactivates the compound and makes it lesser active.
a) I, II
b) I
c) III
d) None
Q.3 Fentanyl can be synthesized from 4-N-propanoylanilinopiperidine by N-alkylation with?
a) 2-phenylethychloride
b) 2-ethylchloride
c) 3-methoxybutane
d) Aniline
Q.4 Side effects of drug Fentanyl is/are?
a) Asthenia
b) Anorexia
c) Apnoea
d) All of the above
Q.5 Match the following drugs with their correct molecular weight.
| i. Fentanyl | A. 228.5 gm/mol |
| ii. Estazolam | B. 336.5 gm/mol |
| iii. Butabarbital | C. 398.3 gm/mol |
| iv. Glycopyrollate | D.212.25 gm/mol |
a) i-A, ii-D, iii-C, iv-B
b) i-B, ii-A, iii-D, iv-C
c) i-A, ii-C, iii-D, iv-B
d) i-D, ii-C, iii-A, iv-B
Q.6 An example of drug from class Opioid analgesic is?
a) Orphinadrine
b) Alprazolam
c) Fentanyl
d) Flavoxate
Q.7 The type of ring system found in the structure of fentanyl is?
a) Piperidine
b) Furan
c) Cyclohexane
d) No ring structure present
Participate in Online FREE GPAT TEST: CLICK HERE
Participate in Online FREE Pharmacist TEST: CLICK HERE
Participate in Online FREE Drug Inspector TEST: CLICK HERE
ANSWERS
1-c
2-d
3-a
4-d
5-b
6-c
7-a