FLUFENAMIC ACID Synthesis, SAR, MCQ, Structure, Chemical Properties and Therapeutic Uses
Flufenamic acid
IUPAC nomenclature
2-{[3-(Trifluoromethyl)phenyl]amino}benzoic acid
Classification
- NSAID
- Anthranylic acid derivatives
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 281.23 g/mol |
| 2 | Physical appearance | Solid |
| 3 | Melting point | 133.5°C |
| 4 | Solubility | 9.09 mg/L |
| 5 | Octanol/water partition coefficient | 5.25 |
| 6 | Presence of ring | Phenyl |
| 7 | Number of chiral centers | Not present |
Mechanism of Action
- Flufenamic acid is a COX inhibitor and prevents the formation of prostaglandins.
- It binds with prostaglandins F synthatase and reduces the activity of enzyme.
- Due to this, it acts as an anti-inflammatory drug and also reduces the pain.
Structure Activity Relationship
General SAR for anthranylic acid derivatives can be summarized as follows:
- Activity decreases when substitution on anthranilic acid ring.
- Activities due to substitution on the N-aryl ring follows the general order m > o > p.
- For disubstitution products, activity was found to be maximum when o and m positions are substituted near to each other on the N-aryl ring.
- Substitutions on the N-aryl ring with such groups which leads the ring to be noncoplanar with the anthranilic acid ring increases the binding of the drug and hence, increases the activity (meclofenamic acid being more active than flufenamic acid).
- NH-moeity of the anthranilic ring is important for the activity of the drug, and replacement of NH- moiety with O, CH3, S, SO2, N-CH3 or N-COCH3 groups decreases the activity of drug.
- Position of the acidic function is important for the activity and not the nature of acidic function. [1]
- Replacement of carboxylic acid function with isosteric tetrazole function has no significant effect on the activity of compound.[1]
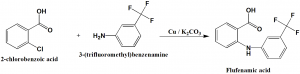
Method of synthesis
Flufenamic acid can be synthesized by the reaction of 2-chlorobenzoic acid and 3-trifluoromethylaniline in the presence of potassium carbonate and copper fillings. [2]
Therapeutic Uses
Flufenamic acid is used for:
- Short-term treatment for mild to moderate pain
- Dysmenorrhea
Side Effects
Side effects of Flufenamic acid are:
- Skin rash
- Changes in vision
- Shortness of breath
- Weight gain
- Swelling
- Stomach bleeding
- Nausea
- Jaundice
- Pale skin
- Difficulty in urination
- Anemia
- Skin reactions
- Indigestions
- Nausea
- Tremors
- Stomach pain
- Confusion
- Diarrhea
- Constipation
- Drowsiness
- Dizziness
- Headache
MCQs
Q.1 Match the following with correct SAR of the class Anthranylic acid derivatives NSAIDs-
| i. Substitution on anthranilic acid ring | A. Increases the activity |
| ii. Meclofenamic acid is | B. Decreases the activity |
| C. More active than Flufenamic acid | |
| D. Less active than Flufenamic acid |
a) i-A, ii-C
b) i-A, ii-D
c) i-B, ii-C
d) i-B, ii-D
Q.2 Correct sequence for the True/False for correct IUPAC names of the drug can be?
- Flufenamic acid: 2-{[3-(Trifluoromethyl)phenyl]amino}benzoic acid
- Auranofin: gold(1+);(3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxane-2-thiolate
- Cytarabine: 17-allyl-7,8-didehydro-4,5α-epoxymorphinan-3,6α-diol.
- Thiobarbital: 5,5-diethyl-2-thioxodihydropyrimidine-4,6(1H,5H)-dione
a) TFTF
b) FFFF
c) TTTT
d) TFFT
Q.3 Molecular weight of drug Flufenamic acid is?
a) 281.23 gm/mol
b) 458.6 gm/mol
c) 406.21 gm/mol
d) 162.1 gm/mol
Q.4 Flufenamic acid mechanism of action includes?
a) It Reduces inflammation
b) Synthesis of prostaglandins is reduced
c) COX is inhibited
d) All of the above
Q.5 Which amongst the following is a therapeutic use of drug Flufenamic acid?
a) Treatment of gout
b) Reducing pain
c) Bronchospasm
d) All of the above
Q.6 Which of the following drug and their classification are correct?
I. Neostigmine: ß-Adrenergic blocker
II. Flufenamic acid: Anthranylic acid NSAID
III. Enflurane: Phenothiazine antipsychotics
IV. Meperidine: Benzodiazepine sedative-hypnotic
a) III, IV
b) I, II, III
c) I, II, III, IV
d) II
Q.7 Flufenamic acid can be synthesized by the reaction of 3-(fluoromethyl)benzamine with?
a) 3-chlorobenzaldehyde
b) 2-chlorobenzoic acid
c) 2-chlorobutanoic acid
d) Oxalic acid
Participate in Online FREE GPAT TEST: CLICK HERE
Participate in Online FREE Pharmacist TEST: CLICK HERE
Participate in Online FREE Drug Inspector TEST: CLICK HERE
ANSWERS
1-c
2-d
3-a
4-d
5-b
6-d
7-b