THIAMYLAL Synthesis, SAR, MCQ,Structure,Chemical Properties and Therapeutic Uses
Thiamylal
IUPAC nomenclature
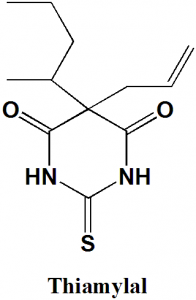
5-(Pentan-2-yl)-5-(prop-2-en-1-yl)-2-sulfanylidenedihydropyrimidine-4,6(1H,5H)-dione
Classification
Thiamylal is a barbiturate sedative-hypnotic.
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 254.35g/mol |
| 2 | Physical appearance | Solid |
| 3 | Melting point | 132-133°C |
| 4 | Octanol/water partition coefficient | 3023 |
| 5 | Solubility | Water solubility is 3.48e-04 M |
| 6 | Presence of ring | Pyrimidine |
| 7 | Number of chiral centers | 2 |
Mechanism of Action
i. Drug binds with different binding sites associated with chloride ionopore at the GABAA
ii. This results in increase in the duration of time for the opening of the chloride ionopore.
iii. As a result, the post synaptic inhibitory effect of GABA in the thalamus is prolonged.
Structure Activity Relationship
- Tri-keto form is most stable in aqueous solution.
- 4,6-dialcoholic tautomeric forms are least stable in aqueous solution.
- 5,5-disubstituted barbituric acid is the prime requirement for the barbiturates to be sedative hypnotics.
- Esterification of either of the 1,3-diazine nitrogens decreases hypnotic activity.
- Substitution of either of the 1,3-diazine nitrogens with aliphatic carbons retains the anticonvulsive properties.
- Esterification of the 5th-position substituents yields agents with analgesic activity but with weak hypnotic properties.
- Introduction of the polar functional group at the 5th– position yields compounds which are fully devoid of sedative-hypnotic or anticonvulsive activity.
- As the number of carbons at R2 carbon increases, the lipophillicity of the drug increases.
- Modification of the 2nd-position oxygen of the barbiturate backbone with sulfur atom yields thiobarbiturate derivatives with increased lipophillicity, shorter duration of action, faster time of onset compared to oxy-derivative. [1]
Method of synthesis
Thiamylal can be synthesized by condensation of diethyl 2-allyl-2-(pentan-2-yl)malonate with thiourea.
Therapeutic Uses
Thiamylal is used for:
- Induction in surgical anesthesia
- Anticonvulsant to counteract the side effect of other anesthetics
- Inducing drowsiness
- Reducing the psychological excitement
- Reducing anxiety.
Side Effects
Side effects of Thiamylal are:
- Insomnia
- Dizziness
- Drowsiness
- Loss of coordination
- Nausea
- Vomiting
- Constipation
- Restlessness
- Headache
- Loss of appetite
- Pale skin
- Shallow breathing
MCQ
Q.1 Due to Potentiation of GABA receptors by Thiamylal?
a) Increase in opening duration of calcium channels
b) Increase in opening duration of the Chloride channels
c) Increase in opening duration of the Sodium channels
d) Decrease in opening duration of the calcium channels
Q.2 Therapeutic use of drug Thiamylal is/are?
a) Induction of surgical anesthetics
b) Inducing drowsiness
c) Reducing anxiety
d) All of the above
Q.3 Which amongst the following are the correct statements with respect to the SAR of drug Thiamylal
I. Tri-keto form is most stable in aqueous solution.
II. 4,6-dialcoholic tautomeric forms are least stable in aqueous solution.
III. 5,5-disubstituted barbituric acid is the prime requirement for the barbituares to be sedative hypnotics.
IV. Esterification of either of the 1,3-diazine nitrogens decreases hypnotic activity.
a) I, III, IV
b) II, IV
c) I, II, III
d) I, II, III, IV
Q.4 The starting chemicals required for the synthesis of drug Thiamylal?
a) Malonic ester
b) Barbital
c) Both a) and b)
d) None of the above
Q.5 Correct sequence for the True/False for the physiochemical properties of the drug Thiamylal is?
I. Molecular weight is 254.35 g/mol
II.It is present in liquid crystalline form
III. Melting point is 132.5oC
IV. No chiral carbons are present in the structure of barbital.
a) TFTF
b) TFFF
c) FFFT
d)FTTF
Q.6 Correct statements for the IUPAC nomenclatures of the drugs are?
I. Thiamylal: 5-(Pentan-2-yl)-5-(prop-2-en-1-yl)-2-sulfanylidenedihydropyrimidine-4,6(1H,5H)-dione.
II, Secobarbital: 5-(pentan-2-yl)-5-(prop-2-en-1-yl)-1,3-diazinane-2,4,6-trione
III. Quazepam: 5-butan-2-yl-5-ethyl-1,3-diazinane-2,4,6-trione
IV. Triazolam: 5-Ethyl-5-phenyl-1,3-diazinane-2,4,6-trione
a) II, III
b) II, IV
c) I, II, IV
d) I, II
Q.7 Match the following drugs with their correct classifications-
| i. Thiamylal | A. Barbiturate sedative-hypnotic |
| ii. Zolpidem | B.Benzazepine |
| iii. Olanzapine | C. Nonbenzodiazepine agonist |
| iv. Oxazepam | D. Benzodiazepine |
a) i-A, ii-C, iii-B, iv-D
b) i-B, ii-D, iii-A, iv-C
c) i-A, ii-D, iii-C, iv-B
d) i-C, ii-A, iii-B, iv-D
Participate in Online FREE GPAT TEST: CLICK HERE
Participate in Online FREE Pharmacist TEST: CLICK HERE
Participate in Online FREE Drug Inspector TEST: CLICK HERE
ANSWERS
1-b
2-d
3-d
4-a
5-a
6-d
7-a
REFERENCES
[1] Lemke TL, Zito SW, Roche VF, Williams DA. Essentials of Foye’s principles of medicinal chemistry. Wolters Kluwer; 2017, 490-91