TIOTIXENE or THIOTHIXENE Synthesis, SAR, MCQ,Structure,Chemical Properties and Therapeutic Uses
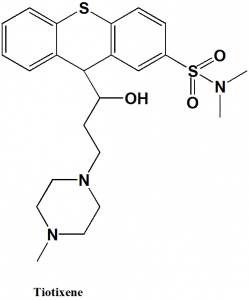
Tiotixene or Thiothixene
IUPAC nomenclature
(9Z)-N,N-dimethyl-9-[3-(4-methylpiperazin-1-yl)propylidene]-9H-thioxanthene-2-sulfonamide.
Classification
Tiotixene is a thioxanthene antipsychotic drug.
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 443.6 g/mol |
| 2 | Physical appearance | White to tan crystalline powder |
| 3 | Melting point | 114-118°C |
| 4 | Solubility | Practically insoluble in water |
| 5 | Octanol/water partition coefficient | 3.78 |
| 6 | Presence of ring | Piperazine, thioxanthene |
| 7 | Number of chiral centers | 2 |
Mechanism of Action
Tiotixene hydrochloride acts as antagonist for following postsynaptic receptors:
- Dopaminergic receptors subtypes D1, D2, D3 and D4, which results in antipsychotic properties on productive and unproductive symptoms.
- Serotonergic receptors 5-HT1 and 5-HT2, which is responsible for the anxiolytic, antidepressive and antiaggressive properties and also as an attenuation of extrapyramidal side effects of the drug.
- Histaminergic receptors H1 subtype, which is responsible for sedation, antiemesis, vertigo, lowering of blood pressure and weight gain properties of the drug.
- α1 and α2 receptors which are responsible for the properties of drug like antisympathomimetic, lowering of blood pressure, reflex tachycardia, vertigo. Sedation. Hypersalivation and incontinence and also sexual dysfunctions; may also attenuate peudoparkinsonism.
- Muscarinic M1 and M2 receptors which causes anticholinergic symptoms like dry mouth and blurred vision.
Structure Activity Relationship
Structure activity relationship of phenothiazine like compounds can be described as follows:
- Tilting of side chain towards ring A grants favorable Vander Waal’s interaction of the side chain. This interaction decides the potency of the drug towards the dopamine receptors.
- Optimal neuroleptic activity occurs when the ring A substituent is in the 2nd-position.
- A trifluoromethyl substituent provides a greater number of favorable Van der Waal’s contacts with the side chain than the chlorine substituent. Thus, phenothiazne with trifluoromethyl substituents are more potent than those with chlorine substituent.
- A piperazine side chain provides more Van der Waal’s contacts with 2-substituent than the alkylamino side chain. Thus, piperizine phenothiazine are more potent in antischizophrenic effects than alkylamino phenothiazines.
- Hydroxyethylpiperazine side chain phenothiazines displays more favorable Van der Waal’s interactions with ring A than simple piperazines.
- In the thioxanthene and xanthenes containing ring systems, the cis forms are more potent neuroleptics than the trans isomers.
- Phenothiazine analogues having the presence of exolytic double bond are more potent than the corresponding compounds lacking the exolytic double bonds. [1]
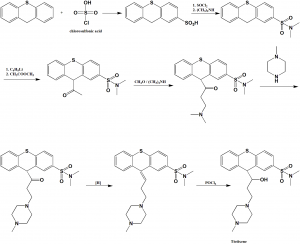
Method of synthesis
i. 9H-thioxantene is reacted with chlorosulfonic acid to give 9H-thioxanten-2-sulfonic acid.
ii. On reaction with thionyl chloride and dimethylamine, the product transforms into 2-dimethylaminosulfonyl-9H-thioxantene.
iii. On reaction of the above formed compound with butyllithium and then with methyl acetate gives the product 9-acetyl-2-dimethylaminosulfonyl-9H-thioxantene.
iv. Aminomethylation of the resulting product with dimethylamine and formaldehyde gives 9-(2-dimethylamineopropionyl)-2-dimethylaminosulfonyl-9H-thioxantene.
v. On reaction with 1-N-methylpiperazine lead to the substitution of the dimethylamine group with N-methyliperazine group.
vi. On reduction of the carbonyl group of the product to secondary hydroxyl group using sodium borohydride, followed by dehydration with the help of phosphorus oxychloride produces the desired tiotixene [2]
Therapeutic Uses
Tiotixene is used for:
- Treatment of schizophrenia
- Reducing nervousness
- Reducing aggression
- Decreasing hallucinations
Side Effects
Side effects of Tiotixene are:
- Dizziness
- Drowsiness
- Lightheadedness
- Constipation
- Blurred vision
- Dry mouth
- Trouble sleeping
- Stiff muscles
- Muscle spasms
- Twisting neck
- Arching back
- Eyes rolling up
- Depression
- Suicidal thoughts
MCQs
Q.1 What can be the correct IUPAC nomenclature of Tiotixene?
a) (9Z)-N,N-dimethyl-9-[3-(4-methylpiperazin-1-yl)propylidene]-9H-thioxanthene-2-sulfonamide.
b) (9Z)-N,N-dimethyl-9-[3-(4-methylpiperazin-1-yl)propylidene]-9H-thioxanthene-2-sulfonic acid
c) 3-(2-chlorophenothiazin-10-yl)-N,N-dimethylpropan-1-amine;hydrochloride
d) N,N-dimethyl-3-phenothiazin-10-ylpropan-1-amine;hydrochloride
Q.2 Which amongst the following statements is/are INCORRECT related to the SAR of tiotixene?
I. A piperazine side chain provides more Van der Waal’s contacts with 2-substituent than the alkylamino side chain. Thus, piperizine phenothiazine are more potent in antischizophrenic effects than alkylamino phenothiazines.
II. Hydroxyethylpiperazine side chain phenothiazines displays more favorable Van der Waal’s interactions with ring A than simple piperazines.
III. In the thioxanthene and xanthenes containing ring systems, the cis forms are more potent neuroleptics than the trans isomers.
IV. Phenothiazine analogues having the presence of exolytic double bond are more potent than the corresponding compounds lacking the exolytic double bonds.
a) I, III, IV
b) I, II
c) III, IV
d) None
Q.3 Number of chiral carbons present in the structure of tiotixene?
a) 0
b) 1
c) 2
d) 3
Q.4 Side effects of drug Tiotixene is/are?
a) Blurred vision
b) Insomnia
c) Muscle spasms
d) All of the above
Q.5 Match the following drugs with their correct melting points:
| i. Tiotixene | A. 116oC |
| ii. Thiopental | B.190oC |
| iii. Favipiravir | C. 159oC |
| iv. Secobarbital | D.100oC |
a) i-A, ii-C, iii-B, iv-D
b) i-C, ii-A, iii-D, iv-B
c) i-C, ii-A, iii-B, iv-D
d) i-B, ii-D, iii-A, iv-C
Q.6 An example of drug from class Thioxanthene antipsychotic drug is?
a) Donepezil
b) Promazine
c) Tiotixene
d) Both a) and b)
Q.7 The type of ring system found in Tiotixene?
a) Phenothiazine
b) Piperizine ring
c) Thioxanthene ring
d) Both b) and c)
Participate in Online FREE GPAT TEST: CLICK HERE
Participate in Online FREE Pharmacist TEST: CLICK HERE
Participate in Online FREE Drug Inspector TEST: CLICK HERE
ANSWERS
1-a
2-d
3-c
4-d
5-a
6-c
7-d