Concept of Somatic Hybridization, Somatic Embryogenesis, and its Applications, MCQ for GPAT, GATE, & CSIR NET
Somatic hybridization
Concept
Sexual hybridization is a conventional method used to improve characteristics in the cultivated plant. Sexual hybridization can be only be used for the same plant species.
In Somatic hybridization, a hybrid cell is formed by In-vitro protoplast fusion. Then this fused protoplast is developed to form a hybrid plant. Plant protoplast has provided novel approaches, to create a new genetically modified cell. Hence, Plant protoplasts can be used in genetic manipulations or modification and improvement of the crop for somatic cells. Protoplast fusion can be used for different plant species, hence somatic hybridization has an advantage over sexual hybridization.
Steps involved in somatic hybridization
- Protoplast fusion
- Hybrid cells selection
- Hybrid plant identification
1. Protoplast fusion
- The protoplast has no cell wall, hence it very easy to fuse two different protoplasts of the different genome. As these two protoplasts have no cell wall there is no barrier for the incompatibility between species.
This fusion of protoplast is done by three methods
- Spontaneous fusion: In this process, homokaryocytes are formed, when there is a fusion of two protoplasts isolated during enzymatic degradation from adjacent cells. Hence, it is a natural process, where fused protoplast may contain varying numbers of nuclei from 2 to 40. Spontaneous fused protoplast does not regenerate into the whole plant.
- Mechanical fusion: In this method, two protoplasts are forced to fuse in enzyme solutions. However, there are chances of protoplast damage during the process.
- Induced fusion: In this method, two protoplasts are fused by using fusion-inducing agents or called as fusogens. Two protoplasts of different strains undergo fusogen treatment, which causes adhesion of protoplasts, leading to the formation of fused protoplast of heterokaryon and then to form a hybrid. eg. of fusogens: polyethylene glycol, polyvinyl alcohol, lysozyme, dextran, fatty acids, esters, NaNO3, high pH/Ca2+ and by electrofusion, etc.
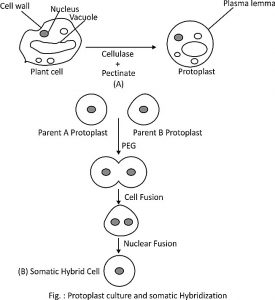
2. Hybrid cell selection
Generally, 20-25% of the protoplast is involved in the infusion process. Alter fusion is completed, the fused protoplasts contain homokaryons (unfused chloroplasts) and heterokaryons among the total protoplast population. Hence it is necessary to identify hybrid cells or so-called heterokaryon. This is done by biochemical method, cytometric methods, and visual method.
3. Hybrid cell identification
This process is required for the genetic contribution of the parental protoplast to develop hybrid cells and then to develop a hybrid plant. Identification can be done by molecular techniques like PCR, counting the number of chromosomes, isoenzymes analysis of hybrid plants, etc.
Cybrids or cytoplasmic hybrids
Cybrids, in which, the cytoplasm is derived from both parents and the nucleus from one parent. The phenomenon for the formation of Cybrids is known as cybridization. Cybrids are hybrids only for cytoplasm.
Applications of somatic hybridization
- It has wide applications in developing disease-resistant plants e.g. tobacco plant resistant against tobacco mosaic virus.
- Genes responsible for environmental tolerance can be introduced in plants to develop environment tolerance. e.g. development of cold tolerance in tomatoes
- The quality characteristics of the plant can be done by somatic hybrids e.g. production of high nicotine content and low erucic content in tobacco.
- The study of cytoplasmic genes can be done by somatic hybrids.
- Somatic hybridization can be done in juvenile plants also.
Limitations
- It is not always possible to produce plants that give visible or fertile seeds
- Genetic instability is associated with protoplast culture
- Diploid protoplast is not recommended for protoplasmic fusion
- It is difficult to produce viable somatic hybrids by protoplasmic fusion.
- Selection Method selection of hybrids are not always efficient
Somatic embryogenesis
Concept
Somatic embryogenesis is one of the micropropagation techniques or clonal propagation. In, Clonal propagation genetically identical copies of individual plants are multiplicated, hence it is an asexual reproduction process. Asexual reproduction is done by the multiplication of vegetative parts. This is only a method for in-vivo propagation. Somatic embryogenesis is the asexual process of regeneration of embryos from organs, tissues, and somatic cells. Non-zygomatic embryos or somatic embryos formed from somatic organs, Parthogenetic embryos formed from an unfertilized egg, and androgenic embryos are formed by somatic embryogenesis.
Somatic embryos are the same as sexually formed or zygotic embryos, which can be excised from parent tissues and induced to germinate into culture media.
Somatic embryos are done by culturing somatic cells. The cells used are epidermis, parenchymatous cells of secondary root phloem, or parenchymatous cells of petioles.
Somatic embryogenesis can be done by two methods
- Direct somatic embryogenesis: In this method, somatic embryos are developed in explants without callus formation. This direct formation of the embryo is due to pre-embryonic determined cells (PEDC) present in some tissues of the plant.
- Indirect somatic embryogenesis: In this method, the first callus if formed by the proliferation of cells from the explant. These cells are used to form cell suspension cultures. Induced embryogenic determined cells (IEDC) from cell suspension culture can form somatic embryos. Embryogenesis is done by certain growth regulators with suitable environmental conditions. In this method, a large number of embryos can be generated in a small volume of culture medium. Hence, the indirect method is commercially used, where somatic embryos formed have good regeneration capability and are synchronous.
Advantages of micropropagation
- Only a piece of plant tissue is required to grow a large number of plants.
- Production of pathogen-free plants can be done by micropropagation
- It is a cost-effective process, as it requires very little space and the cost of production is very low.
Multiple-choice Questions
1. Which of the following can be done in different plant species
a) Somatic embryogenesis
b) Protoplast fusion
c) Somatic hybridization
d) All
2._____________is the advantage of somatic hybridization over sexual hybridization
a) It can be done in the same plant
b) It can be done in the same plant species
c) It can be done in different plant species
d) All
3. First step in somatic hybridization is ________
a) Identification of hybrid plant
b) Protoplast fusion
c) Protoplast isolation
d) Identification of hybrid cell
4. Hybrid cell is made by fusing two protoplast in__________
a) Callus culture
b) Suspension culture
c) Gene cloning
d) Somatic hybridization
5. _________ are used for protoplast fusion
a) Chemical agents
b) Fusogens
c) Vitamins
d) Plant hormones
6. ______________is the fusogenic agent
a) Polyvinyl alcohol
b) Ethyl alcohol
c) Propranolol
d) Methanol
7. The following are the methods used in hybrid cell selection EXCEPT:
a) Cytometric method
b) Biochemical method
c) Visual method
d) By PCR
8. Plant characteristics can be improved by __________
a) Callus culture
b) Suspension culture
c) Sexual hybridization
d) Somatic hybridization
9. Somatic embryogenesis is based on___________
a) Sexual reproduction
b) Asexual reproduction
c) Both
d) None
10. Which of the following type of plant cells can be used for somatic embryogenesis
a) Palisade cells of leaves
b) Epidermis
c) Parenchymatous cells of xylem
d) Medullary cells of stem
Answer Key
- d
- c
- c
- d
- b
- a
- d
- d
- b
- b
Reference
- Satyanarayana, biotechnology, 1st e.d. Kolkata, books & allied (p) ltd publishers, 2005 page: 528-537 and 557-559