PENTAZOCINE Synthesis, SAR, MCQ,Structure,Chemical Properties and Therapeutic Uses
Pentazocine opioid analgesics
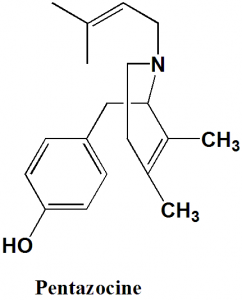
IUPAC nomenclature
2-dimethylallyl-5,9-dimethyl-2′-hydroxybenzomorphan
Classification
- Pentazocine falls under category of opioid analgesics.
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 285.4 g/mol |
| 2 | Physical appearance | White to very tan crystalline powder |
| 3 | Melting point | 145.4-147.2°C |
| 4 | Solubility | Soluble in acetone; sparingly soluble in ethyl acetate |
| 5 | Octanol/water partition coefficient | log Kow = 3.31 |
| 6 | Presence of ring | Piperidine, phenyl |
| 7 | Number of chiral centers | 1 |
Mechanism of Action
Pentazocine antagonizes the opioid effects by competing at the receptor site. It mainly compete the opioid mu receptor site.
Structure Activity Relationship
SAR for morphine analogues can be summarized as follows:
- Replacement of phenolic hydroxyl into –OCH3/-OC2H5 will make the compound less analgesic and cough suppression will also takes place.
- Replacement of alcoholic hydroxyl with –OCH3 makes the compound 5 times more active.
- Replacement of alcoholic hydroxyl with -OC2H5 makes the compound 2.4 times more active than morphine.
- Replacement of alcoholic hydroxyl with –OCOCH3 will also activates the compound by 4.2 times.
- Replacement of alcoholic hydroxyl with ketone group inactivates the compound and makes it lesser active.
- By hydrogenation of alicyclic unsaturated linkage, activity increases by 1.2 times.
- On replacement of the methyl group from tertiary nitrogen by hydrogen atom, activity decreases.
- On replacement of N-CH3 by NCH2CH2Ph, activity increases by 14 times.
- When the methyl group of tertiary nitrogen replaced by N-allyl/methallyl/propyl, the compound so formed acts like the Morphine antagonist.
- When the methyl group of tertiary nitrogen replaced by N(CH3)2 Cl– , compound have curare action and it do not possesses any analgesic activity.
Method of synthesis
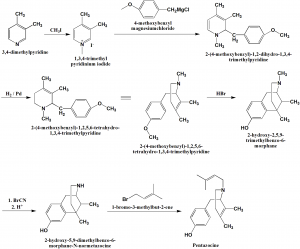
i. 3,4-dimethylpyridine undergoes reaction with methyl iodide to produce 1,3,4-trimethylpyridinium iodide.
ii. The above formed compound is then reacted with 4-methoxybenzyl magnesium chloride to give 2-methoxybenzyl-3,4-dimethyl-1,2-dihydropyridine.
iii. Double bond of the last formed compound is hydrogenated using palladium catalyst to produce 3-methoxybenzyl-3,4-dimethyl-1,2,3,4-tetrahydropyridine.
iv. Intramolecular alkyation and simultaneous demethylation of the resulting compound leads to formation of 2-hydroxy-2,5,9-trimethylbenzo-6-morphane.
v. Above formed compound undergoes N-demethylation by cyanogens bromide to form 2-hydroxy-5,9-dimethylbenzo-6-morphane-N-normetazocine.
vi. On alkylation of the resulting product by 1-bromo-3-methyl-2-butene gives pentazocine.[1]
Therapeutic Uses
Pentazocine is used for:
- Relieve moderate to severe pain
Side Effects
Side effects Pentazocine are:
- Dizziness
- Drowsiness
- Constipation
- Tiredness
- Lightheadedness
- Stomach pain
- Rash
- Difficulty urinating
- Mood changes
- Stomach upset
MCQs
Q.1 Choose the correct option related with the mechanism of action of drug Pentazocine?
a) Acts as Opiate receptor antagonist
b) Mainly Competes with opioid sigma receptors
c) Mainly competes with opioid kappa receptors
d) All of the above
Q.2 Therapeutic use of drug Pentazocine is/are?
a) Treatment of diarrhea
b) As an anesthetic
c) Relieve moderate to severe pain
d) All of the above
Q.3 Which amongst the following are the correct statements with respect to the SAR of drug Pentazocine?
I. Replacement of alcoholic hydroxyl with -OC2H5 makes the compound 2.4 times more active than morphine.
II. Replacement of alcoholic hydroxyl with –OCOCH3 will also activates the compound by 4.2 times.
III. Replacement of alcoholic hydroxyl with ketone group inactivates the compound and makes it lesser active.
IV. By hydrogenation of alicyclic unsaturated linkage, activity increases by 1.2 times
a) I, IV
b) II, III
c) III, IV
d) I, II, III, IV
Q.4 Number of chiral carbons present in the structure of Pentazocine is?
a) 0
b) 1
c) 2
d) 3
Q.5 Correct sequence for the True/False for the physiochemical properties of the drug pentazocine is?
I. Molecular weight = 582 gm/mol
II. Melting point = 110.2oC
III. White to very tan crystalline powder
IV. Ring structure absent
a) FFTF
b) TFFT
c) FTTF
d) TFFF
Q.6 Correct statements for the IUPAC nomenclatures of the drugs are?
I. Pentazocine: 2-dimethylallyl-5,9-dimethyl-2′-hydroxybenzomorphan
II. Mechlorethamine: 2-Chloro-N-(2-chloroethyl)-N-methylethan-1-amine
III. Ifosfemide: N,3-Bis(2-chloroethyl)-1,3,2-oxazaphosphinan-2-amide 2-oxide
IV. Carmustine: 1,3-Bis(2-chloroethyl)-1-nitrosourea
a) I, II
b) I, II, III, IV
c) II , III
d) III, IV
Q.7 Match the following drugs with their correct classifications-
| i. Ifosfemide | A. Nitrogen mustard |
| ii. Pentazocine | B. Nitrosoureas alkylating agent |
| iii. Lomustine | C. Pyrimidine antagonist |
| iv. Cytarabine | D. Opioid analgesic |
a) i-B, ii-D, iii-C, iv-A
b) i-B, ii-C, iii-A, iv-D
c) i-C, ii-B, iii-D, iv-A
d) i-A, ii-D, iii-B, iv-C
Participate in Online FREE GPAT TEST: CLICK HERE
Participate in Online FREE Pharmacist TEST: CLICK HERE
Participate in Online FREE Drug Inspector TEST: CLICK HERE
ANSWERS
1-a
2-c
3-d
4-b
5-a
6-b
7-d
REFERENCES
[1] Vardanyan R, Hruby V. Synthesis of essential drugs. Elsevier; 2006 Mar 10.