PHENINDAMINE Synthesis, SAR, MCQ,Structure,Chemical Properties and Therapeutic Uses
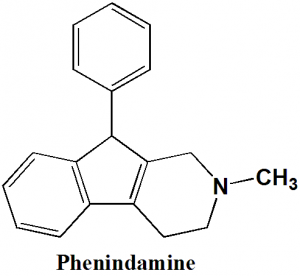
Phenindamine
IUPAC nomenclature
2-methyl-9-phenyl-2,3,4,9-tetrahydro-1H-indeno[2,1-c]pyridine
Classification
- H1-receptor antihistamine
- Alkylamine antihistamine
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 261.4 g/mol |
| 2 | Physical appearance | Solid |
| 3 | Melting point | 91oC |
| 4 | Solubility | 2.77e-02 g/L |
| 5 | Octanol/water partition coefficient | N/A |
| 5 | Presence of ring | Indenopyridine, phenyl |
| 6 | Number of chiral centers | 1 |
Mechanism of Action
- Phenindamine competes with histamine for binding with H1 receptor site on the effector cell.
- Thus, all those pharmacological responses which were mediated by histamine at H1-receptor site are reduced and the intensity of allergic reactions and tissue injury response involving histamine release are reduced.
Structure Activity Relationship
Structure activity of alkyl amines antihistamines can be summarized as:
- E- and Z- isomers in alkenes shows large difference in activity, where, E-isomers are more potent than Z-isomers.
- The two aromatic rings have different binding environments at the receptors.
- 5-6 angstrom distance is required between aromatic ring and tertiary aliphatic amine for biding at the receptor.
- S-enantiomers have greater affinity for H1 histamine receptors [1]
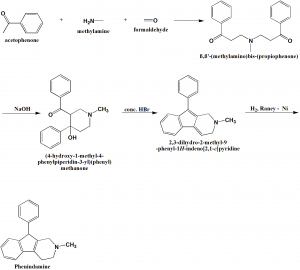
Method of synthesis
- Acetophenone, methylamine and formaldehyde are reacted together to get ß,ß’-(methylamino)bis-(propiophenone).
- The above formed compound is treated with sodium hydroxide to get 3-benzoyl-4-hydroxy-1-methyl-4-phenyl-piperidine.
- On reaction of last with concentrated hydrogen bromide, 2-methyl-9-phenyl-2,3-dihydro-1H-indeno[2,1-c]pyridine is formed.
- The last is reduced to get phenindamine. [2]
Medicinal Uses
Phenindamine is used for treatment of:
- Allergies
- Hay fever
- Common cold
- Watery eyes
- Runny nose
- Sneezing
Side Effects
Side effects of Phenindamine are:
- Fatigue
- Dizziness
- Drowsiness
- Sleepiness
- Dry mouth
- Enlarged prostate
- Difficulty in urination
- Headache
- Difficulty breathing
- Closing of throat
- Swellings in face
MCQs
Q.1 Match the following with correct SAR of the alkylamines antihistamine drugs:
| i. E-isomers are | A. More potent than Z-isomers |
| ii. S-enantiomers have | B. Less potent than Z-isomers |
| C. Greater affinity for H1-receptors | |
| D. Greater affinity for H2-receptors |
- i-A, ii-C
- i-A, ii-D
- i-B, ii-C
- i-B, ii-D
Q.2 Correct sequence for the True/False for correct IUPAC names of the drug can be?
- Phenindamine: 2-methyl-9-phenyl-2,3,4,9-tetrahydro-1H-indeno[2,1-c]pyridine
- Diphenhydramine: 2-(diphenylmethoxy)-N,N-dimethylethanamine
- Auranofin: gold(1+);3,4,5-triacetyloxy-6-(acetyloxymethyl)oxane-2-thiolate;triethylphosphanium
- Meloxicam: 4-hydroxy-2-methyl-N-(5-methyl-1,3-thiazol-2-yl)-1,1-dioxo-1λ6,2-benzothiazine-3-carboxamide
a) TFFT
b) FFTF
c) TTTT
d) FFFT
Q.3 Molecular weight of Phenindamine is?
a) 200.5 gm/mol
b) 540.6 gm/mol
c) 261.4 gm/mol
d) 1042 gm/mol
Q.4 Phenindamine binds with which receptor to produce antihistaminic actions?
a) α-receptors
b) ß-receptors
c) Nicotinic receptor
d) H1-receptor
Q.5 Which amongst the following is not a therapeutic use of drug phenindamine?
a) Runny nose
b) Sneezing
c) Hay fever
d) Hypertension
Q.6 Which of the following drug and their classification are correct?
I. Phenindamine: H1 receptor antihistamine drug
II. Cimetidine: H2 receptor antihistamine drug
III. Lansoprazole: Proton pump inhibitor
IV. Thiotepa: Antineoplastic drug
a) I, III
b) I, II, III, IV
c) III, IV
d) I, II, IV
Q.7 Number of chiral centers present in the structure of phenindamine is?
a) 0
b) 1
c) 2
d) 3
Participate in Online FREE GPAT TEST: CLICK HERE
Participate in Online FREE Pharmacist TEST: CLICK HERE
Participate in Online FREE Drug Inspector TEST: CLICK HERE
Participate in CSIR NET JRF Mock Test
ANSWERS
1-a
2-c
3-c
4-d
5-d
6-b
7-b
REFERENCES
[1] Lemke TL, Williams DA, editors. Foye’s principles of medicinal chemistry. Lippincott Williams & Wilkins; 2012 Jan 24.
[2] US 2 470 108 (Roche; 1949; appl. 1947).