Types of Plant Culture: Callus Culture, Suspension culture, Batch culture. MCQ for DBT BET,GPAT, GATE, & CSIR NET
Callus Culture
The undifferentiated and unorganized, plant cell mass is called a callus. Callus contain differentiated and non-differentiated tissues. Callus cells are parenchymatous, the mature cells or non-dividing cells undergo changes to achieve a meristematic state or cell division and lead to the formation of callus growth is called dedifferentiation. These meristematic cells have the capability to grow into the whole plant this phenomenon is known as redifferentiation. This dedifferentiation and redifferentiation are two components of cellular Totipotency.
Requirements of callus culture
- Explants: explants may be root, leaf, stem, flower, anther, etc, cell division, and multiplication depend on the stage of cell division of explant. If the explant used has meristematic cells then cell multiplication and cell division is rapid.
- Physical requirements: An adequate temperature of 22-28o darkness and light depends on plant species.
- Growth regulators: Growth regulators in nutrient media depends on the explant selected its genotype, its endogenous content of hormone, etc. generally Auxins and cytokinins are used as growth regulators in culture media for callus culture.
Simple Steps involved in callus culture
- Selection of explant
- Sterilization of explant
- The explant is inoculated into Agar media
- The explant is then subcultured to form Callus
- The callus developed is then transformed into a liquid medium
- These callus in liquid medium forms clumps and results in suspension culture
Applications of Callus culture
- It is used for somaclonal variation, genetic transformation
- It is used for the production of secondary metabolites
- For nutritional requirements of the plant
- Differentiation of plant cells and organ
- Used for the development of protoplast and suspension culture.
Suspension culture
It is a cell culture technique used for cell division and development of the whole plant from the plant cell. Generally, leaves are used to obtain a homogenous population of Cells used for Invitro culture. These cells can be obtained by either mechanical method i.e. by mechanically grinding the surface-sterilized leaves in a homogenizer. The mixture is centrifuged at low speed to remove debris. The supernatant is used to achieve the required cells. The enzymatic method in which, under suitable osmotic pressure macerozyme is used to release cells from leaf tissues, which degrades lamella and cell walls of parenchymatous tissues. Cultured tissues like callus culture is also used to isolate single cells by the subculturing method. The single cells isolated by suitable methods are grown in suspension cultures. Subculturing is done in fresh media, to maintain cell suspensions. The cells incubated in suspension culture, cells get enlarged and start dividing. The incubation period depends on initial cell density, the growth rate of cells, and the duration of the lag phase (preparation of cell to divide). Suspension culture requires normally 21-28 days for incubation.
Batch culture
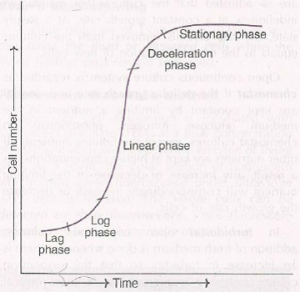
It is a type of suspension culture, in which cells are grown in a fixed amount of culture media with required nutrition. As nutrients and O2 supply become limited, cell division, cell growth, and biomass increases. Maintenance of the batch cultures can be easily done by transferring small inoculum into a fresh suspension medium at a regular time of 2-3 days. Cells growth in suspension culture is represented by five phases
- Lag Phase: In this phase, cells prepare to divide
- Log Phase: In this phase, the cell multiplication is at the highest rate, hence also known as the exponential phase.
- Linear phase: In this phase, there is a reduction of cell division and increased cell size expansion
- Deceleration phase: In this phase, there decline in cell expansion and division
- Stationary phase: In this phase, there is a constant, cell size, and a number of cells.
(Picture © U. Satyanarayana, biotechnology, this picture is used for Educational purpose only)
Methods for Assessment of growth of cell cultures
To assess the growth of cells in suspension culture, various methods like Cell counting, packed cell volume, and weight increase can be used. Cell counting: This is done by disrupting the cells by treating with pectinase or chromic acid, separated and counted by using a hemocytometer. It is an accurate method although it is tedious and time-consuming. Packed cell volume: In this method, the measured volume of cell suspension is centrifuged at 2000 xg for 5 minutes. The volume of packed cells is recorded. Cell dry weight can be measured by discarding the supernatant and then, washing or cleansing and drying the pellet for overnight. Cell fresh weight: In this method, the suspension is filtered with a preweighed nylon fabric filter with a funnel to collect wet cells. These cells are washed, drained under vacuum, and weighed. This method requires a large amount of samples for accuracy.
Measurement of the viability of cultured cells
Cell viability is an important factor for the growth of the cells. It can be done by using stains and observing under a microscope. Phase-contrast microscopy: viability of the cells can be checked by the presence of health nuclei. Evan’ blue staining: In this, the cells are stained with diluted Evan’s blue of 0.025% W/V dye. Only dead or damaged cells can be dyed by the stain and the viable cells remain unstained. Fluorescein diacetate method (FDA): in this method the cell suspension is incubated with FDA of 0.01% concentration. Esterase enzyme of Viable or living cells Cleaves FDA, by which the polar portion of fluorescein is released. This polar portion of fluorescein emits green fluorescence in UV light. Hence the viable cells can be detected by their fluorescence due to the accumulation of the polar portion of fluorescein.
Limitations of cell cultures
- Cultured cells and not genetically stable and hence they may have a chance to undergo mutation.
- The damage of cells is more frequent. This can be prevented by vigorous stirring to prevent aggregation.
- Strict aseptic conditions have to be followed to avoid contamination.
Applications of Cell culture
- Cellular metabolic pathways can be elucidated by using cell cultures.
- Cell culture serves as a good target for the production of secondary metabolites for commercial use.
- For crop improvement
- Desirable mutants or mutations can be done by cultured cells.
Multiple Choice Questions
1. _______types of cells are required to facilitate cell division a) Meristematic cells b) Palisade cells c) Parenchymatous cells d) Chollenchymatous cells 2. ______ is the requirements of callus culture a) Explants b) Culture media c) Only A d) A & B 3. Auxins are required for ________ a) Callus culture b) Suspension culture c) Both d) None 4. The following are the methods for assessment for growth of cells EXCEPT: a) Cell counting b) Packed cell volume c) Cell fresh weight d) PCR 5. ___________is the method for assessment of viability of cells a) Cell counter method b) Packed cell volume C) Phase contrast microscopy d) Cell fresh weight 6. ____________is the method to differentiate viable and non-viable cells a) FDA method b) Evan’s blue stain c) Both d) None 7. Centrifugation is done for ________method to measure growth of cells a) Packed cell volume b) Cell weight c) Only A d) None 8. Cell size expansion is maximum in _______phase of batch culture a) Lag phase b) Log phase c) Linear Phase d) Stationary phase 9. In batch culture cell division and cell growth depends on _____________ a) High O2 b) Low oxygen c) Nutrients d) None 10. Genetic Mutation is one of the limiting factor in _______ a) Protoplast culture b) Cell culture c) Callus culture d) All Answer Key
- a
- d
- c
- d
- c
- c
- c
- c
- b
- b
Participate in Online FREE GPAT TEST: CLICK HERE
Participate in Online FREE Pharmacist TEST: CLICK HERE
Participate in Online FREE Drug Inspector TEST: CLICK HERE
Participate in CSIR NET JRF Mock Test
Participate GATE Mock Test Reference
- Satyanarayana, biotechnology, 1st e.d. Kolkata, books & allied (p) ltd publishers, 2005 page: 500-503