CARBAMAZEPINE Synthesis, SAR, MCQ, Structure, Chemical Properties and Therapeutic Uses
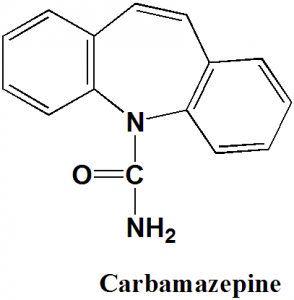
Carbamazepine
IUPAC nomenclature
5H-dibenzo[b,f]azepine-5-carboxamide
Classification
Carbamazepine belongs to class anticonvulsants.
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 236.27 g/mol |
| 2 | Physical appearance | White to off-white powder. |
| 3 | Melting point | 190.2°C |
| 4 | Solubility | Soluble in alcohol, acetone,; practically insoluble in water. |
| 5 | Octanol/water partition coefficient | 2.77 |
| 6 | Presence of ring | Dibenzazepine ring |
| 7 | Number of chiral centers | Not present |
Mechanism of Action
i. Carbamazepine binds and blocks neuronal sodium channels / voltage sensitive calcium channels.
ii.This results in blockage or suppression of neuronal depolarization and hypersynchronization.
iii. Seizures cause due to hypersynchronization is controlled.
Structure Activity Relationship
SAR tricyclic antidepressants can be summarized as follows:
- Amino group and side alkyl chain do not affect the activity
- Presence of 4 carbons or more on the side chain decreases the activity.
- Methyl substituents on amino groups increases activity.
- Substitution on amino group with ethyl or larger groups increases the toxicity and decreases the activity of drug.
- Removal of the benzene ring makes the drug inactive.
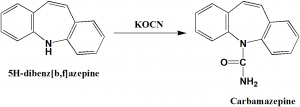
Method of synthesis
Reaction of 5H-dibenz[b,f]azepine with potassium cyanate to get carbamazepine.
Therapeutic Uses
Carbamazepine is used for:
- Prevention and control of seizures
- Providing relieve from certain nerve pains
Side Effects
Side effects of Carbamazepine are:
- Dizziness
- Drowsiness
- Nausea
- Vomiting
- Unsteadiness
- Dry mouth
- Headaches
- Liver problems
- Loss of appetite
- Dark urine
- Mouth sores
- Blurred vision
- Swelling of ankles
- Numbness
- Confusion
MCQs
Q.1 Choose the correct option related with the mechanism of action of drug carbamazepine?
a) It binds and blocks neuronal sodium channels
b) It binds and blocks voltage sensitive calcium channels
c) Neuronal depolarization and hypersynchronization takes place
d) All of the above
Q.2 Therapeutic use of drug carbamazepine is/are?
a) Prevention of seizures
b) Prevention of blood clotting
c) Prevention of Diabetes
d) All of the above
Q.3 Which amongst the following are the correct statements with respect to the SAR of drug carbamazepine?
I. Amino group and side alkyl chain do not affect the activity
II. Presence of 4 carbons or more on the side chain increases the activity.
III. Methyl substituent on amino groups decreases activity.
IV. Substitution on amino group with ethyl or larger groups increases the toxicity and decreases the activity of drug.
a) I, IV
b) II, III
c) II, IV
d) I, III
Q.4 5H-dibenz[b,f]azepine ca be converted into carbamazepine by reaction with?
a) Methanol
b) Potassium cyanide
c) Potassium cyanate
d) Ethanoic anhydride
Q.5 Correct sequence for the True/False for the physiochemical properties of the drug Carbamazepine is?
I. Molecular weight = 136.24
II. Melting point = 190.2oC
III. White to off-white powder
IV. Ring structure absent
a) FFTF
b) TFFT
c) FTTF
d) TFFF
Q.6 Correct statements for the IUPAC nomenclatures of the drugs are?
I. Carbamazepine: 5H-dibenzo[b,f]azepine-5-carboxamide.
II. Loxapine: 1-(Aminomethyl)cyclohexaneacetic acid
III. Gabapentin: 8-chloro-6-(4-methylpiperazin-1-yl)benzo[b][1,4]benzoxazepine
IV. Thiopental: 5-ethyl-5-pentan-2-yl-2-sulfanylidene-1,3-diazinane-4,6-dione
a) I, II
b) II, IV
c) I, III
d) III, IV
Q.7 Match the following drugs with their correct classifications-
| i. Carbamazepine | A. Anxiolytic agent |
| ii. Diazepam | B. Anticonvulsant |
| iii. Buspirone | C. Benzodiazepine Antipsychotic drug |
| iv. Loxapine | D. Benzazepine Antipsychotic drug |
a) i-B, ii-D, iii-C, iv-A
b) i-B, ii-C, iii-A, iv-D
c) i-C, ii-B, iii-D, iv-A
d) i-A, ii-D, iii-A, iv-C
Participate in Online FREE GPAT TEST: CLICK HERE
Participate in Online FREE Pharmacist TEST: CLICK HERE
Participate in Online FREE Drug Inspector TEST: CLICK HERE
ANSWERS
1-d
2-d
3-a
4-c
5-c
6-c
7-b