CHLORCYCLIZINE Synthesis, SAR, MCQ,Structure,Chemical Properties and Therapeutic Uses
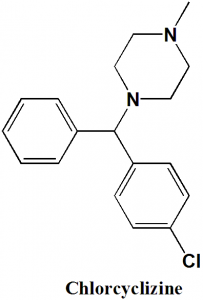
Chlorcyclizine
IUPAC nomenclature
1-[(4-Chlorophenyl)(phenyl)methyl]-4-methylpiperazine
Classification
- H1-receptor antihistamine
- Piperazine antihistamine
Physiochemical Properties
| S. NO. | PHYSICAL AND CHEMICAL PROPERTIES | |
| 1 | Molecular weight | 300.8g/mol |
| 2 | Physical appearance | Oil |
| 3 | Melting point | 216oC |
| 4 | Solubility | Water solubility is 0.0424 mg/ml |
| 5 | Octanol/water partition coefficient | 3.45 |
| 5 | Presence of ring | Piperazine , phenyl |
| 6 | Number of chiral centers | 1 |
Mechanism of Action
Antihistamines such as chlorcyclizine acts as pharmacological antagonists of hitamine at the histamine receptor sites. They do not prevent the release of histamine.
There is also hepatic microsomal enzyme-inducing properties of chlorcyclizine which results in shortening of duration of action of some barbiturates as result of enzyme induction.
Structure Activity Relationship
Structure activity of piperizine antihistamines can be summarized as:
- These are the derivatives of ethylene diamines.
- The connecting moiety is CHN group
- Primary structural difference is nature of para aromatic ring substituent
- These are moderately potent.
- Slow onset of action
- Low incidence of drowsiness
- They also exhibit peripheral and central antimuscarinic activity.
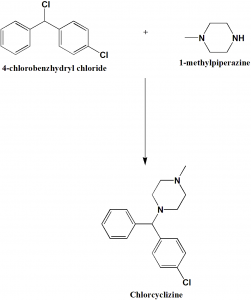
Method of synthesis
Alkylation of 1-methylpiperazine with 4-chlorobenzhydrylbromide gives chlorcyclizine. [1]
Medicinal Uses
Chlorcyclizine has:
- Mildly sedative properties
- Antihistaminic properties
- Slight anticholinergic properties
- Antispasmodic properties
- It enhances the action of epinephrine
- Local anesthetic action
Side Effects
Side effects of chlorcyclizine are:
- Sedation
- Dizziness
- Tinnitus
- Lassitude
- Loss of coordination
- Fatigue
- Blurred vision
- Diplopia
- Euphoria
- Nervousness
- Insomnia
- Tremors
MCQs
Q.1 Mechanism of action of chlorcyclizine drug includes?
a) Release of histamine in prevented
b) Release of histamine is stimulated
c) Antagonistic effects at H1 receptor for histamine
d) Agonist effects H1 receptor for Histamine
Q.2 Therapeutic use of drug Chlorcyclizine is/are?
a) Local anesthetic
b) Sedative
c) Antihistamine
d) All of the above
Q.3 The type of ring present in the structure of chlorcyclizine?
a) Piperazine
b) Pyridine
c) Cyclohexane
d) All of the above
Q.4 Number of chiral carbons present in the structure of Chlorcyclizine?
a) 0
b) 1
c) 2
d) 3
Q.5 Correct sequence for the True/False for the physiochemical properties of the drug Chlorcyclizine can be?
- Molecular weight: 300.8 gm/mol
- Physical appearance: Oil
- Octanol/water partition coefficient: 3.45
a) TTT
b) TFT
c) FFT
d) TFF
Q.6 Correct statements for the IUPAC nomenclatures of the drugs are?
I. Chlorcyclizine: 1-[(4-Chlorophenyl)(phenyl)methyl]-4-methylpiperazine
II. Carbinoxamine: 2-[(4-Chlorophenyl)-pyridin-2-yl-methoxy]-N,N-dimethyl-ethanamine
III. Edrophonium: Ethyl-(3-hydroxyphenyl)-dimethylazanium
IV. Tenoxicam: 4-hydroxy-2-methyl-1,1-dioxo-N-pyridin-2-ylthieno[2,3-e]thiazine-3-carboxamide
a) I, II
b) III, IV
c) IV
d) I, II, III, IV
Q.7 Match the following drugs with their correct classifications-
| i. Chlorcyclizine | A. H2-antagonist |
| ii. Cimetidine | B.Vasodilator |
| iii. Mercaptopurine | C. Antimetabolite |
| iv. Nitroglycerine | D. H1-receptor antihistamine |
a) i-A, ii-C, iii-D, iv-B
b) i-D, ii-A, iii-C, iv-B
c) i-D, ii-B, iii-A, iv-C
d) i-B, ii-D, iii-A, iv-C
Participate in Online FREE GPAT TEST: CLICK HERE
Participate in Online FREE Pharmacist TEST: CLICK HERE
Participate in Online FREE Drug Inspector TEST: CLICK HERE
Participate in CSIR NET JRF Mock Test
ANSWERS
1-c
2-d
3-d
4-b
5-a
6-d
7-b
REFERENCES
[1] US 2 630 435 (Burroughs Wellcome; 1953; prior. 1948).